Expanding Tier 2 Participation Reshapes India’s Trial Ecosystem
The April 2026 edition of IndiaTrialPulse by StriderDCT highlighted continued momentum in India’s multi-site clinical trial landscape, with increasing geographic participation and diversified therapeutic focus.
Built on insights from the Clinical Trials Registry of India (CTRI), the report captured key shifts shaping India’s evolving clinical research environment.
Key Highlights
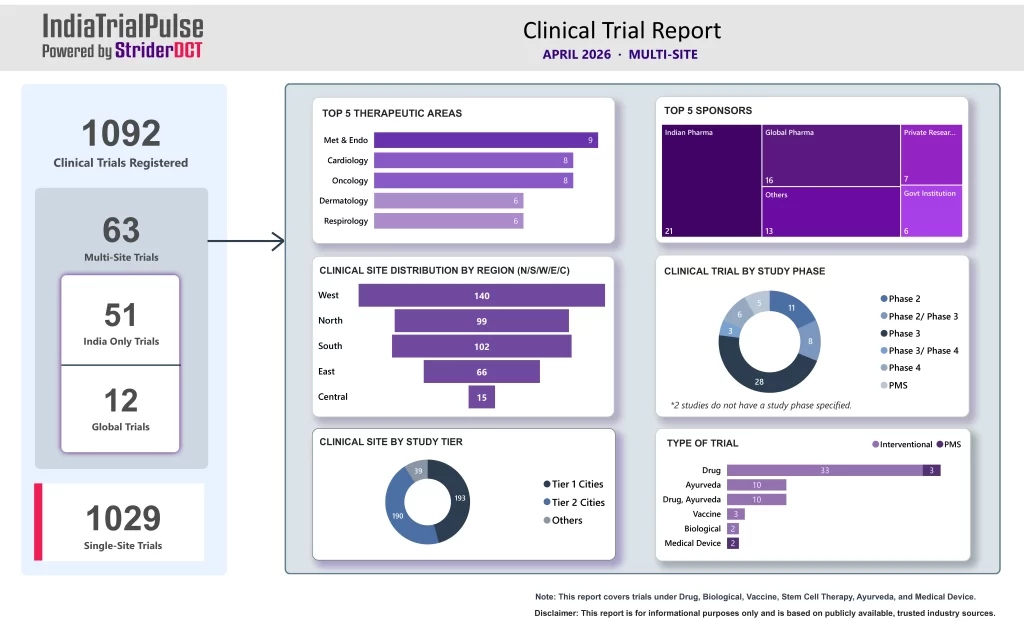
- 63 multi-site trials were registered during April
- Metabolic & Endocrinology, Cardiology, and Oncology led therapeutic activity
- Indian Pharma and Global Pharma remained the dominant sponsor groups
- Tier 1 and Tier 2 city participation was nearly equal
- India-only studies continued to significantly outnumber global trials
What This Indicates
- April demonstrated clear signs of geographic normalization in clinical trial execution, with growing participation beyond traditional metro hubs.
- The balanced contribution between Tier 1 and Tier 2 cities highlights expanding operational reach and increasing readiness for broader decentralized execution models.
- IndiaTrialPulse continues to track the pulse of India’s clinical research ecosystem through structured monthly intelligence.


